Get your daily news on global issues
Provided by AGPCORD FOR LIFE® COMPLETES PHASE 1 DOSING IN FDA AUTHORIZED SACROILIAC JOINT (SIJ) SYNDROME - CLINICAL TRIA
Cord for Life® completes Phase 1 dosing for its FDA-authorized clinical trial using cord blood to treat lower back pain from Sacroiliac Joint (SIJ) Syndrome.
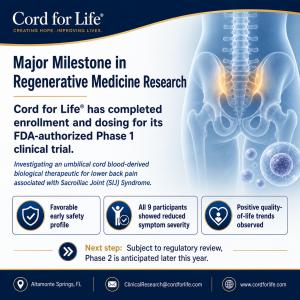
ALTAMONTE SPRINGS, FL, UNITED STATES, May 8, 2026 /EINPresswire.com/ -- ALTAMONTE SPRINGS, FL (May 2026) Cord for Life®, a full service cord blood bank with more than 30 years of experience, today announced the completion of enrollment and dosing in its Phase 1 clinical trial evaluating a biological therapeutic derived from umbilical cord blood under investigation for the treatment of lower back pain arising from the Sacroiliac Joint.The ninth and final subject was injected on January 7, 2026, completing enrollment in the FDA authorized Phase 1a open label, dose ranging study conducted under an active Investigational New Drug (IND) application. The study enrolled nine adult subjects with lower back pain associated with the Sacroiliac Joint.
Phase 1a has demonstrated a favorable early safety profile, with no unexpected adverse events reported to date. Preliminary investigator observations suggest encouraging clinical progress. All participants demonstrated a reduction in symptom severity scores within 3-6 months post-treatment. Favorable trends observed in quality of life and functional status measures within the same timeframe. Subject to completion of required safety follow up and regulatory submissions, we anticipate initiating Phase 2 later this year.
All subjects will continue in the required safety follow-up period over the next twelve months. Completion of the Phase 1 safety evaluation and preparation of the final Clinical Study Report are required prior to initiation of the planned Phase 2 under the existing IND. The University of Florida Data Safety Monitoring Board (DSMB) reports to continue the trial as planned; no protocol modifications are recommended. Based on the current data, the DSMB concludes that the Phase 2 study may proceed without modification. The Phase 2 protocol will be submitted to FDA under the existing IND.
"This marks an important milestone for our clinical development program," said Donald L. Hudspeth, BSCLS, MT (ASCP), General Manager and Director of International Projects for Cord for Life®. "Successful completion of enrollment and dosing in Phase 1 reflects the collaborative efforts of our clinical partners and reinforces our commitment to advancing cord blood derived cellular therapies through a disciplined clinical program and a defined regulatory pathway".
Cord for Life® and its clinical collaborators at the University of Florida in Gainesville under the leadership of Dr. Rene Przkora, MD, PhD, FASA, will continue patient follow up, data collection, and analysis in accordance with FDA requirements. Subject to regulatory review and completion of the required safety period, the company intends to proceed with Phase 2 to further evaluate dosing and expanded enrollment.
PREMIERMAXCB® PLATINUM is an investigational biological therapeutic derived from minimally manipulated umbilical cord blood and manufactured under cGMP conditions. The product is currently limited to clinical investigation under FDA IND authorization.
In addition, our minimally manipulated umbilical cord blood PREMIERMAXCB® products are compliant with the recently passed Florida stem cell law Senate Bill No. 1768 and House Bill No. 1617.
Erin Willigan
Cord for Life
+1 917-207-0234
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.